Why is balancing chemical equations important in chemistry?

Balancing chemical equations helps you understand the relationship between reactants and products in a chemical reaction.
It also helps you satisfy the law of conservation of mass. This law states that mass is neither created nor destroyed — atoms on the reactant and product sides must have the same mass and number.

Learn simple strategies that will have you balancing chemical equations, like putting the same amount of substances on each side of the balance scale.
Did you know?
What you need to know
Chemical equation: a visual representation of a chemical reaction. It has 3 parts.
Reactants: substances present before the chemical reaction.
Products: substances present after the chemical reaction.
Arrow: symbolizes chemical reaction.

Coefficient: numbers placed before the elements or compounds referring to molecules.
Subscript: numbers after the elements or compounds referring to atoms.
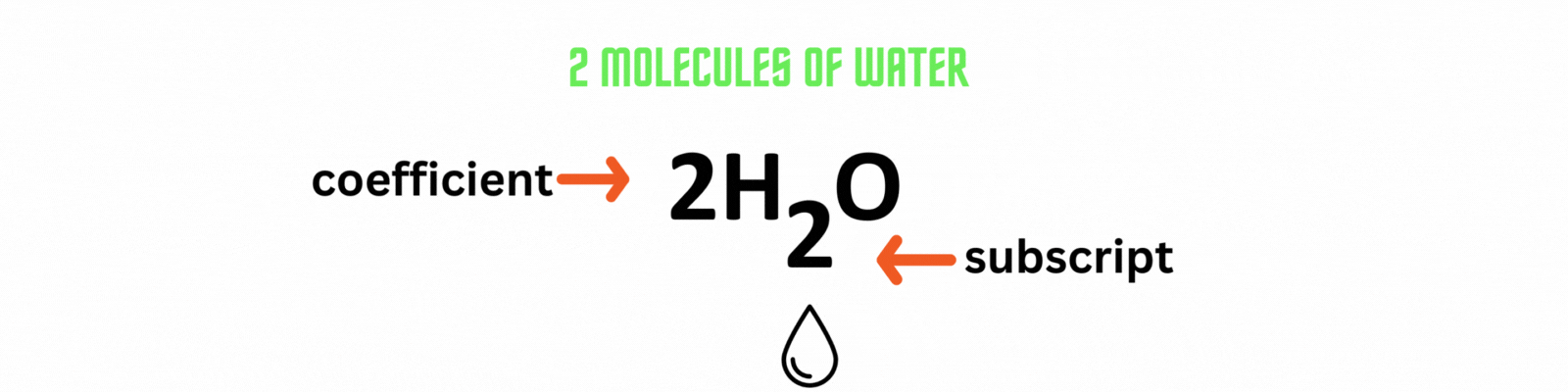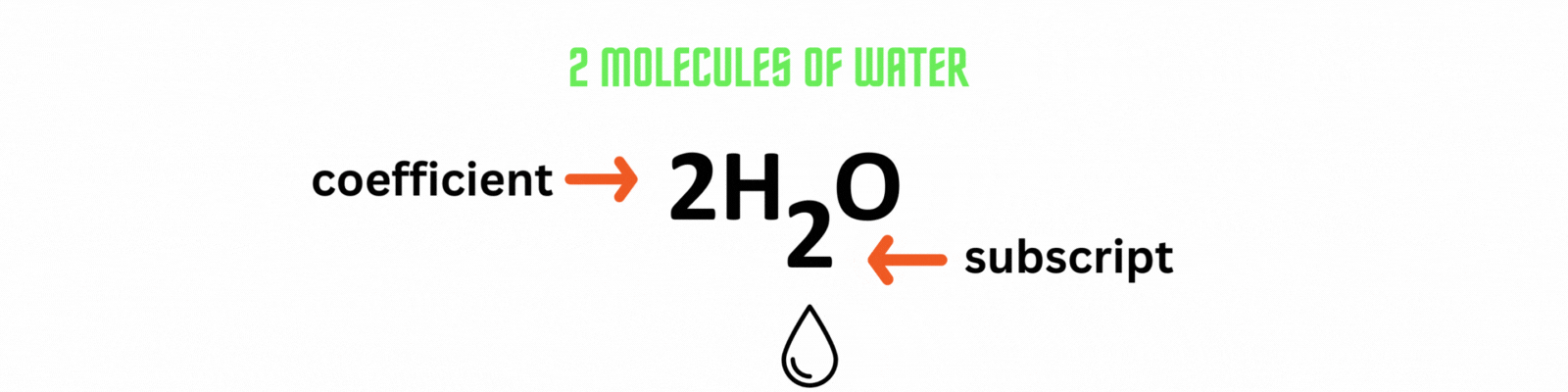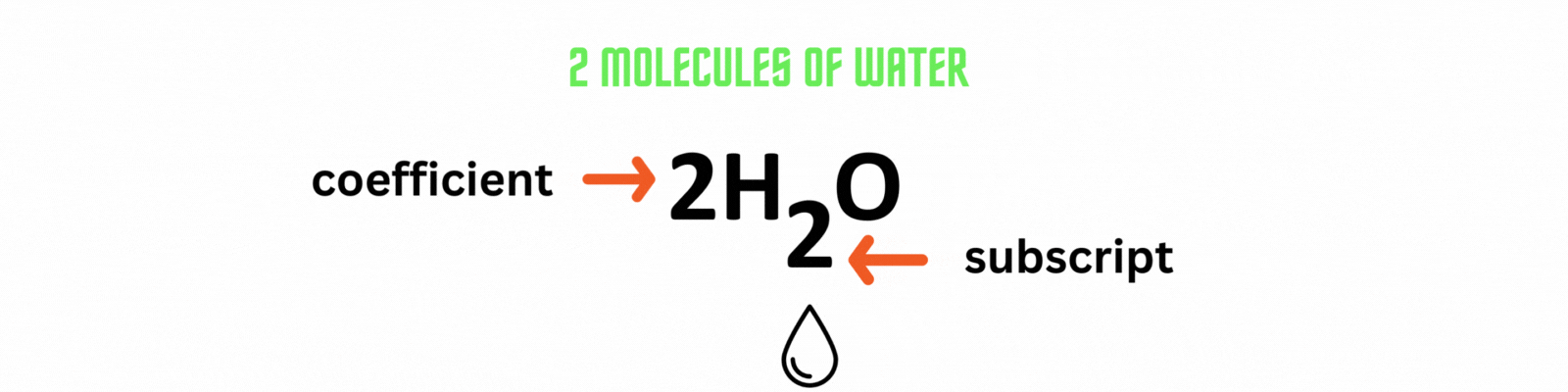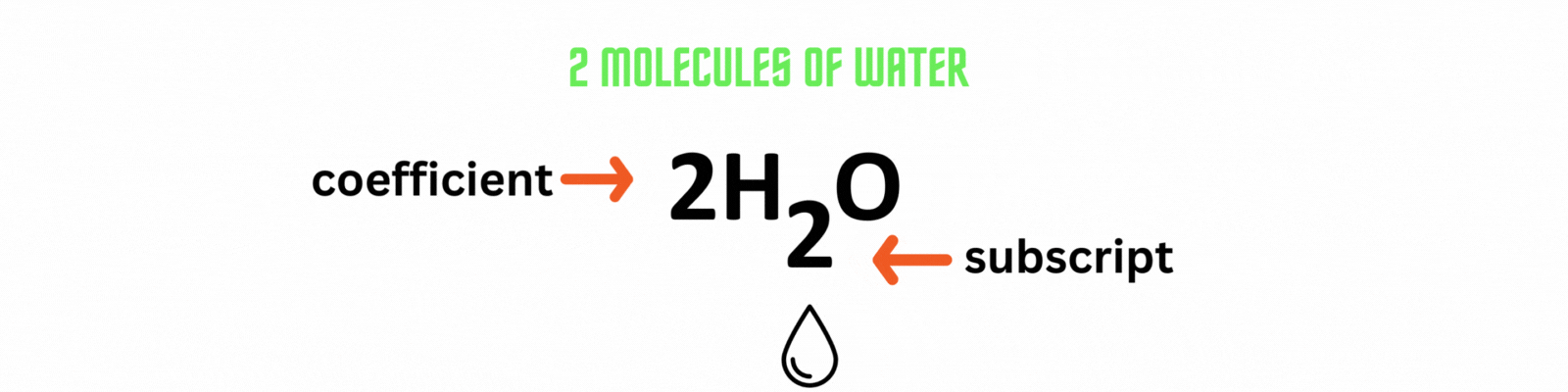
Did you know?
Step 1: Count the number of atoms
Now you're ready to balance a chemical equation! 🙌

Take note of the number of atoms for each element on the reactant and product sides of the chemical equation. ✍🏼
Do's:
Subscript is visible: Count it as the total number of atoms.
Subscript isn't visible: Count it as 1.

Reactants
Carbon atom (C) = 1
Hydrogen atom (H) = 4
Oxygen atom (O) = 2

Products
Carbon atom (C) = 1
Hydrogen atom (H) = 2
Oxygen atom (O) = 2 + 1 = 3

This is NOT a balanced chemical equation! There are unequal numbers of atoms on both sides of the equation.
Quiz
By looking at the chemical equation above, which element(s) have unequal numbers of atoms on reactant and product sides of the equation? Select all that apply:
Did you know?
Step 2: Change the coefficient of the substance(s)

Place the coefficient 2 before the water molecule on the product side then count the number of atoms on each side. ✍🏼
Do's:
Add the coefficient before an element or a compound to balance out a chemical equation. Use the least common multiple between numbers as the coefficient.
Multiply each element's subscript by the coefficient.

Reactants
Carbon atom (C) = 1
Hydrogen atom (H) = 4
Oxygen atom (O) = 2

Products
Carbon atom (C) = 1
Hydrogen atom (H) = 2 x 2 = 4
Oxygen atom (O) = 2 + (2 x 1) = 4

This is NOT a balanced chemical equation yet! There are unequal numbers of oxygen atoms on each side of the chemical equation.
Did you know?
Subscribe for more quick bites of learning delivered to your inbox.
Unsubscribe anytime. No spam. 🙂
Step 3: Repeat until you reach a balanced equation

Place another coefficient 2 before oxygen on the reactant side, then count the number of atoms on each side again. Make sure both the reactant and product sides have the same number of atoms. ✍🏼
Do's:
Use whole numbers when balancing a chemical equation.
Keep changing the coefficient until all elements have the same numbers on both sides. A good rule of thumb is to balance out elements that appear multiple times last. In this equation, you'll balance out oxygen last because it pops up in two molecules on the product side.

Reactants
Carbon atom (C) = 1
Hydrogen atom (H) = 4
Oxygen atom (O) = 2 x 2 = 4

Products
Carbon atom (C) = 1
Hydrogen atom (H) = 2 x 2 = 4
Oxygen atom (O) = 2 + (2 x 1) = 4

This is a BALANCED chemical equation! All number of atoms for carbon (C), hydrogen (H), and oxygen are equal.
Did you know?
Practice your new skill ✍🏼
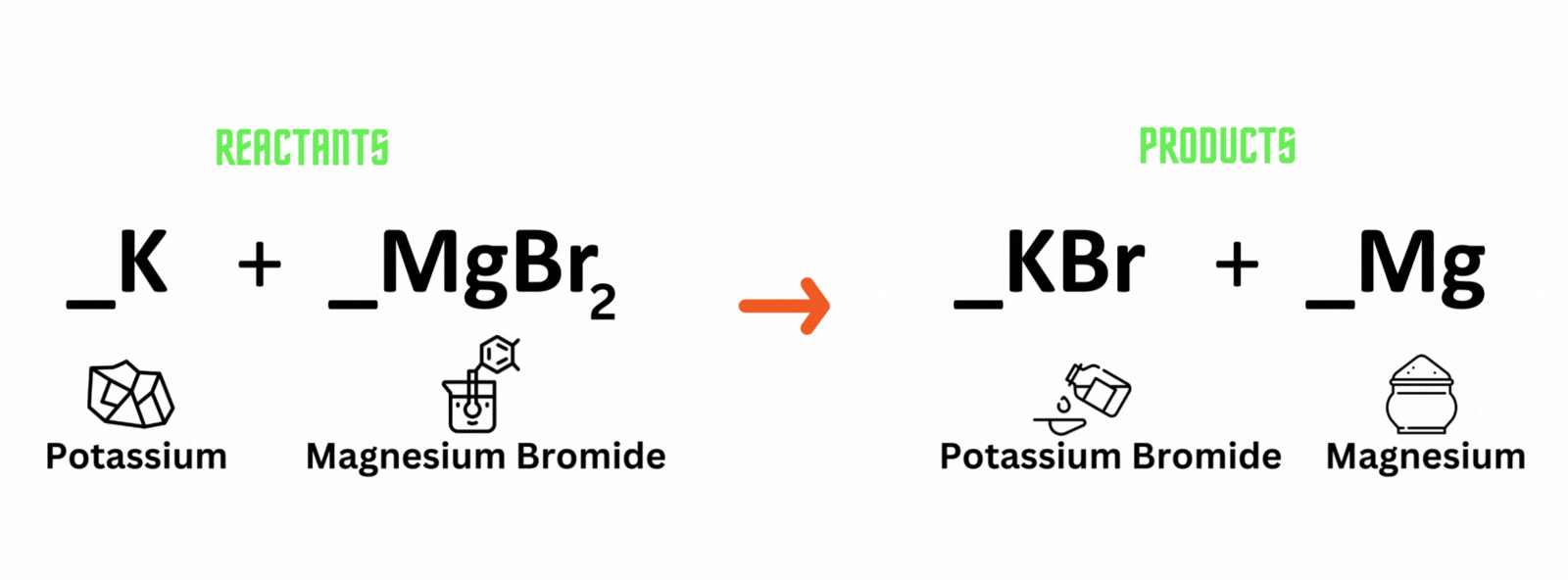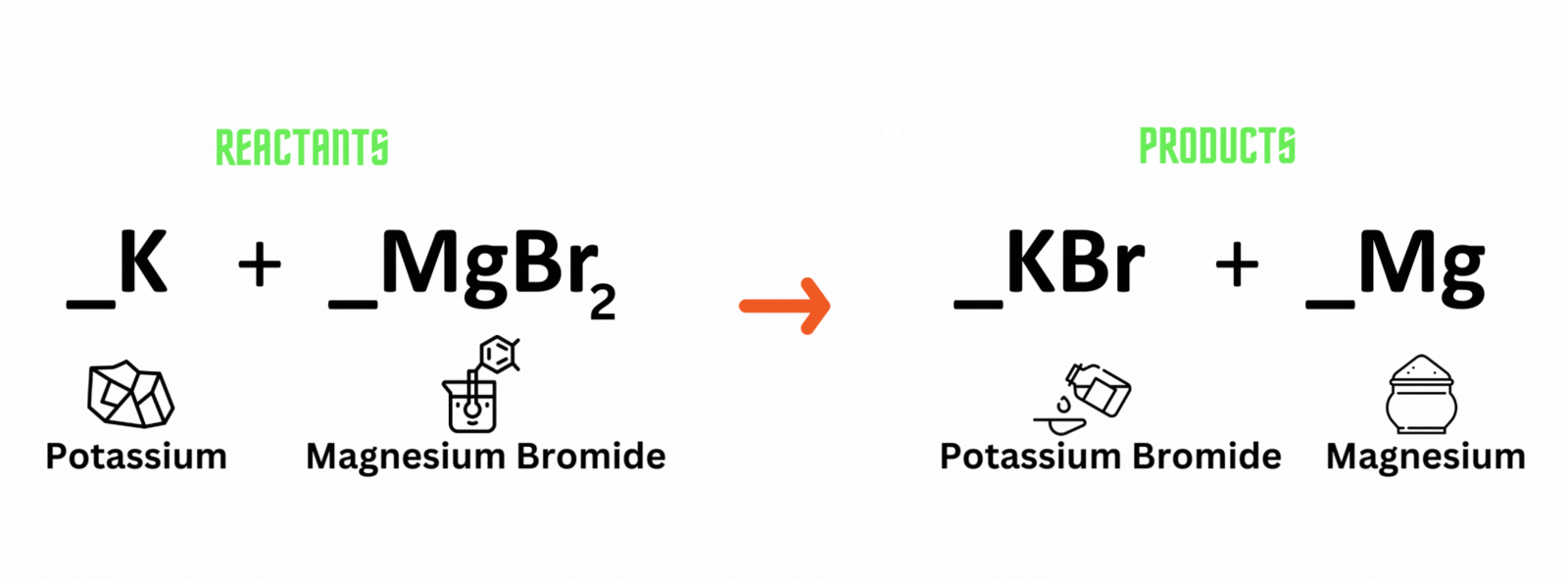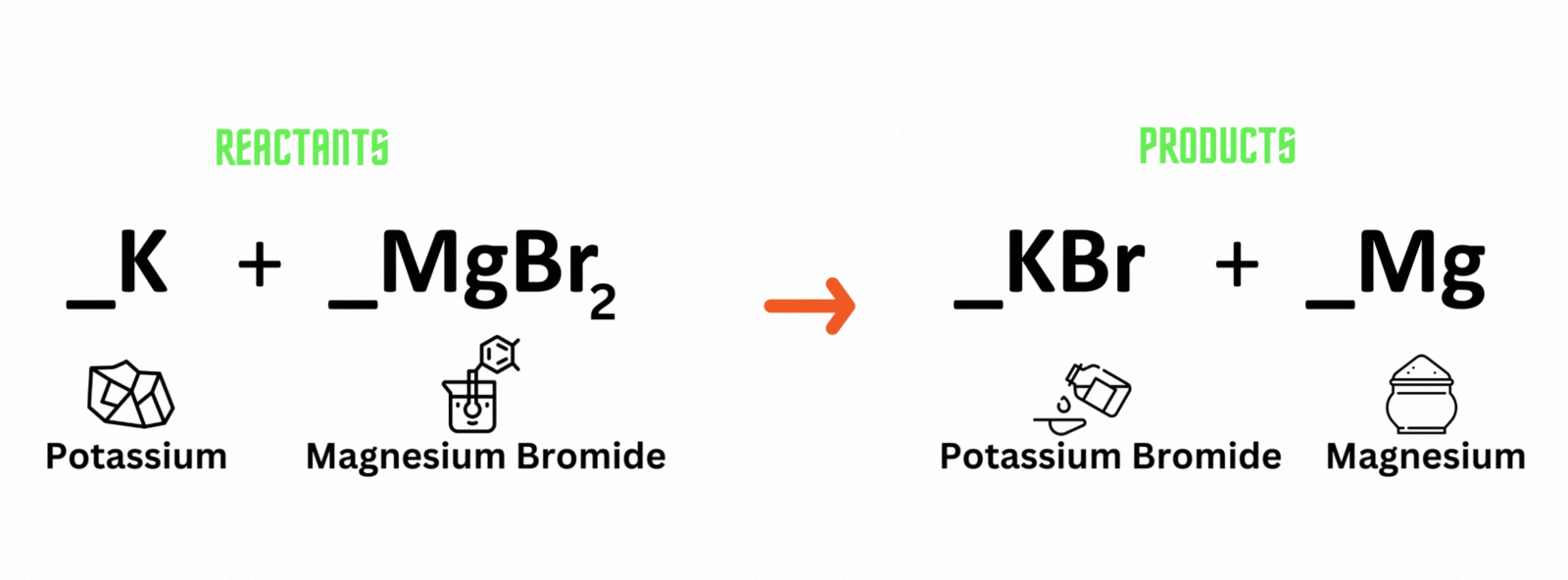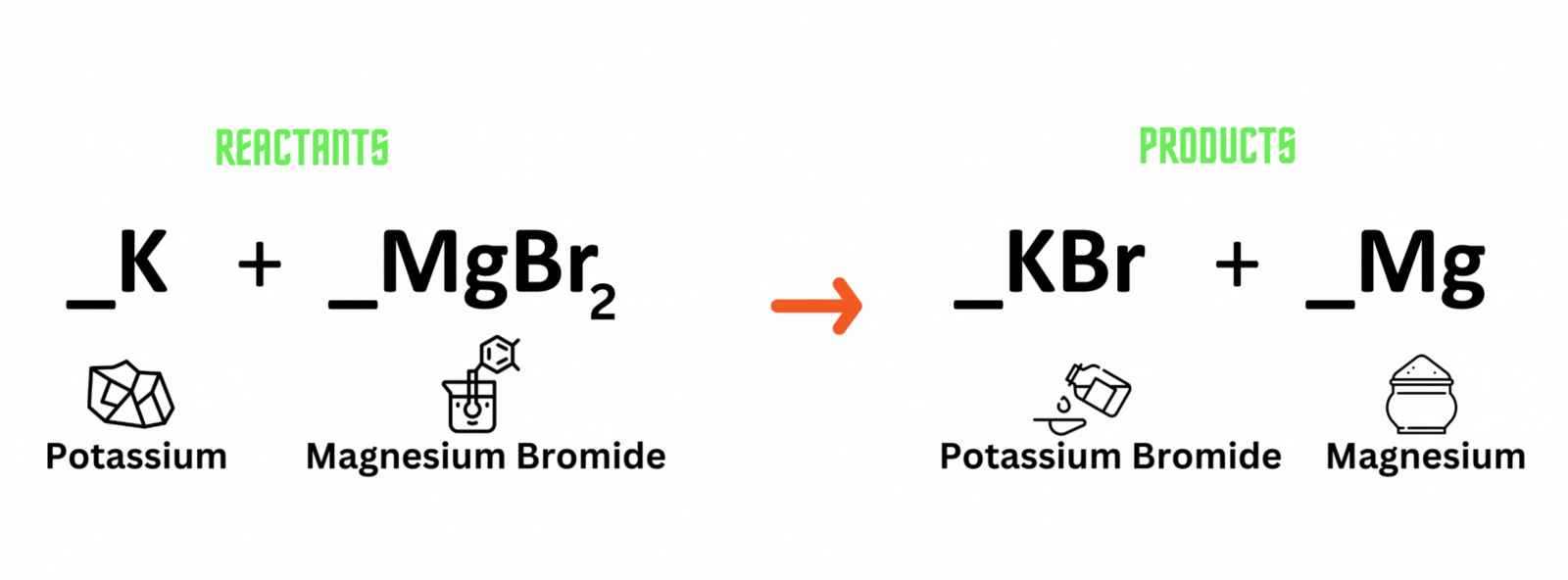
Potassium reacts with magnesium bromide, which produces potassium bromide and magnesium. Where should you place coefficient 2 on the reactant and product sides to balance the equation?
A. Potassium (K) and potassium bromide (KBr)
B. Magnesium bromide (MgBr) and magnesium (Mg)
C. Magnesium bromide (MgBr) and potassium bromide (KBr)
D. Potassium (K) and magnesium (Mg)
Quiz
Which letter above is the best answer?
Take Action

Balancing a chemical equation is a trial-and-error method. Counting the atoms and changing the coefficient until you reach a balanced equation is necessary. Try these activities and polish your new skills:
Your feedback matters to us.
This Byte helped me better understand the topic.
